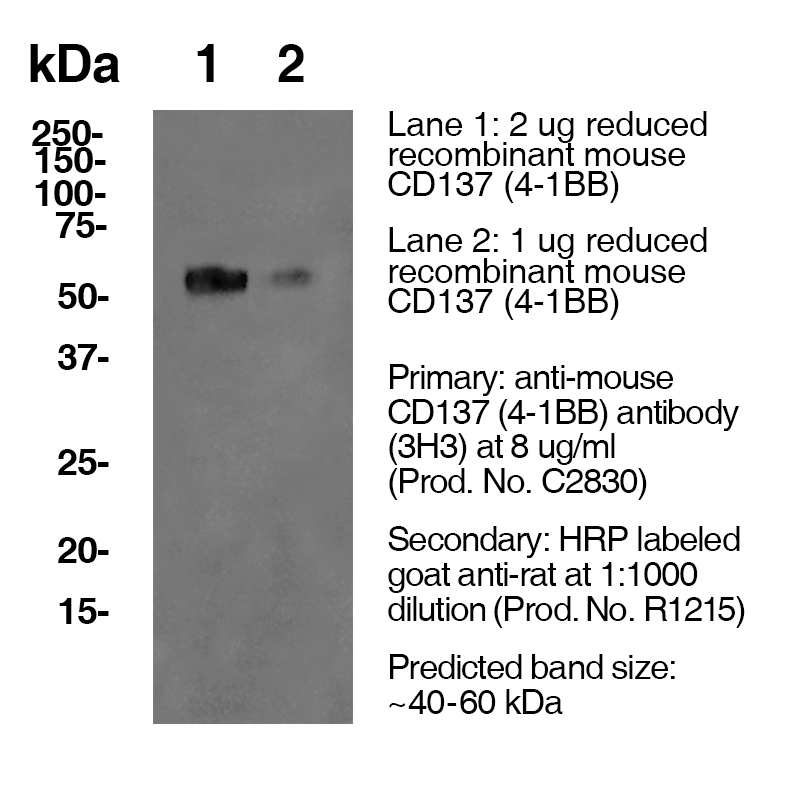
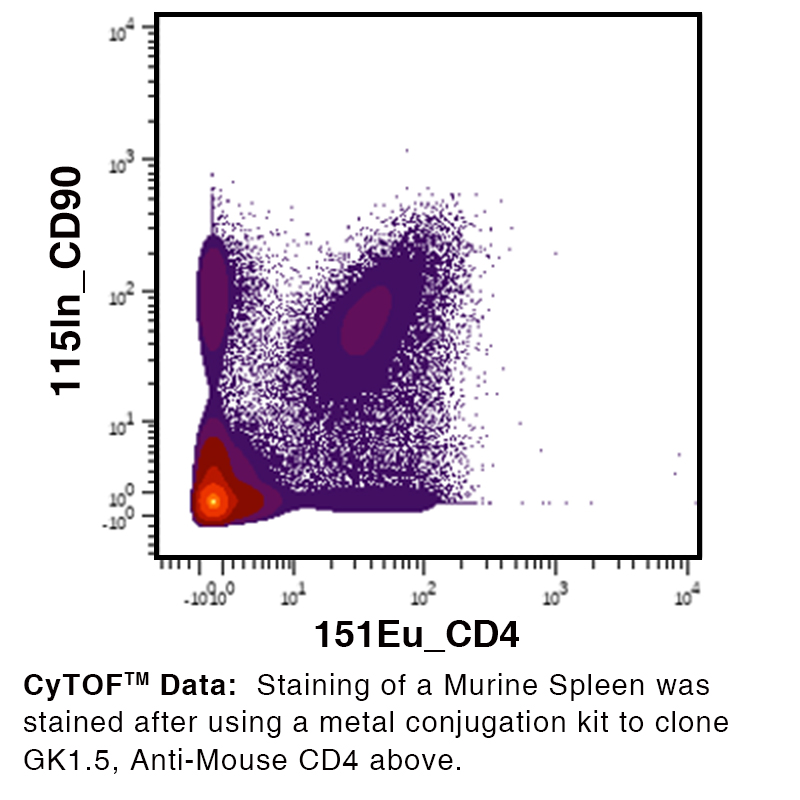
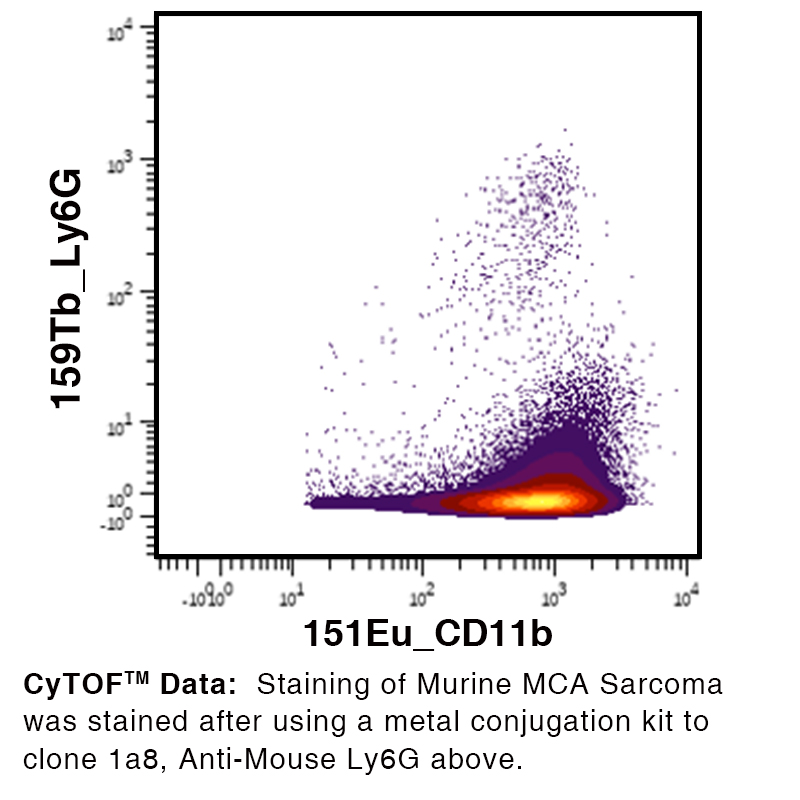
Over 25 Years of Knowledge and Expertise
in Producing Recombinant Antibodies and Conjugates
Custom Antibody Services
Leinco Technologies scientific staff has extensive monoclonal antibody development experience and offers clients the opportunity to collaborate on the development of antibodies against soluble proteins, cell surface molecules, or haptens for use in detection, neutralization or in vivo depletion. Through the use of proprietary development technologies, Leinco has demonstrated success in developing monoclonal antibodies against single amino acid substitutions, splice variants and for neutralization.

Antibody Offerings
- In vivo Functional Grade | Learn More
- Immune Checkpoints | Learn More
- Antibodies for Flow Cytometry | Learn More
- Type I & Type II Interferons | Learn More
- Mouse Cell Depletion | Learn More
- Biosimilars | Learn More
Services Include
- Small - large scale antibody production | Learn More
- Cell Banking and Optimization (MCB & WCB)| Learn More
- Antibody Purification and Bioanalytical Testing | Learn More
- Custom Antibody Conjugation | Learn More
- Antibodies for In Vitro Diagnostics (IVD) | Learn More
- Assay Development | Learn More
Quality Standards
Leinco Technologies operate in a production facility located in St. Louis, Missouri. Cell culture work and bioreactor production are performed in class 10,000 dedicated biopharmaceutical cleanroom suites. Standard operating procedures to ensure high-quality purified antibodies are used. Leinco is an ISO9001.2015 certified company and all manufacturing data is recorded in cGMP compliant batch records.